Activities
- Home
- Activities
- Release
- The role of the hippocampal biological clock in the control of memory recall and its molecular mechanism
Dec 19, 2019
The role of the hippocampal biological clock in the control of memory recall and its molecular mechanism
A paper by the Principal Investigator (A01), Satoshi Kida, has been published in Nature Communications.
Social behaviors including social approach, interaction, and recognition/discrimination are important components to determine and develop personality in human as well as animals. Especially, social recognition memory is an essential and basic component of social behaviors to discriminate familiar and novel animals/humans. However, mechanisms of social memory retrieval are unknown although mechanisms underlying formation of social recognition memory have been extensively investigated.
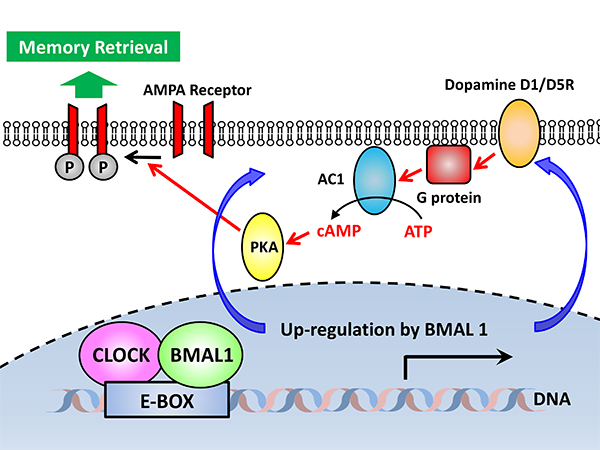
In this current study, Satoshi Kida’s group in the University of Tokyo identified mechanisms for the regulation of social recognition memory retrieval by hippocampal circadian clock at the molecular levels. Cognitive performance in people varies according to time-of-day, with memory retrieval declining in the late afternoon-early evening. According to previous finding in human, Kida’s group investigated roles of hippocampal circadian clock in memory performance by generating and analyzing transgenic mice expressing dominant negative mutant of transcription factor BMAL1 (dnBMAL1), a core circadian clock gene, in forebrain. Interestingly, they found that hippocampal clock controlled by the BMAL1 regulates time-of-day retrieval profile. Indeed, inducible transgenic dnBMAL1 expression in mouse forebrain or hippocampus disrupted retrieval of hippocampal memories at Zeitgeber Time 8-12, independently of retention delay, encoding time and Zeitgeber entrainment cue. Furthermore, they found that this altered retrieval profile was associated with downregulation of hippocampal Dopamine-cAMP signaling in dnBMAL1 mice and that these changes included decreases in Dopamine Receptors (D1-R and D5-R) and GluA1-S845 phosphorylation by PKA. Consistently, pharmacological activation of cAMP-signals or D1/5Rs rescued impaired retrieval in dnBMAL1 mice. Most importantly, GluA1 S845A knock-in mice showed similar retrieval deficits with dnBMAL1 mice. These findings suggest mechanisms underlying regulation of retrieval by hippocampal clock through D1/5R-cAMP-PKA-mediated GluA1 phosphorylation.
Press release
https://www.u-tokyo.ac.jp/focus/en/press/z0508_00081.html
Nature
https://www.nature.com/articles/s41467-019-13554-y
Foreign media reaction
